While some countries are talking about herd immunity, a vaccine for COVID-19 looks like the only possible way to contain the spread. Here is a look at the status & progress of vaccine development across the world.
Globally, the highest ever daily increase in the number of COVID-19 positive cases was recorded on 04 June 2020, when close to 1.3 Lakh new cases were reported. The total number of positive cases globally has crossed 65 lakhs, according to WHO’s dashboard. The number of deaths has crossed 3.9 lakh. Vast majority of the global population is still vulnerable to COVID-19. In India, the number of daily cases has increased substantially ever since the relaxation of lockdown measures. The entire world is waiting for the vaccine to become available. Here is a look at the progress in the development of a vaccine for COVID-19.
Vaccination helps boost immunity against a disease
Scientists and other experts around the world have been working rigorously to develop a vaccine for COVID-19. Vaccination helps a body’s immune system to identify and combat certain pathogens such as virus and bacteria, thereby keeping the person safe from the diseases they cause, some of them which can even be deadly. Through vaccines, the disease-causing germs, which are either killed or weakened such that the person does not get sick, is injected into the body. This stimulates the immune system to produce antibodies which will prevent the disease without actually getting infected.
It can take decades to develop a vaccine
Development of vaccine is a long process and can take years or even decades. Many factors about the infection need to be understood such as its cause – virus or bacteria, how the germs/pathogen infects cells, and how the human immune system will respond to it. The process is long drawn and a complex one which requires the involvement of both public and private organizations. For instance, the first vaccine for the deadly Ebola virus which was discovered around forty-three years ago, was approved by the US Food and Drug Administration, only in December 2019. Now that the COVID-19 pandemic is a global emergency, resources and funding might also be diverted for vaccine development, which will facilitate quicker completion of the vaccine development process.
Vaccine development happens in various stages
There are broadly three stages that vaccine development must undergo, according to WHO. These are developmental, licensure, and post-licensure. The developmental stage can be further sub- classified into preclinical, clinical research and development.
Preclinical research and development are carried out in the laboratory assays and on animals, if necessary. At this stage, information such as details of development and production of vaccine along with reports of control testing are collected, to justify further clinical study in humans.
Clinical trials in humans take place in three phases
Clinical trials in humans are classified into three phases- phase I, phase II, phase III. For this, prior approval is required in some countries like UK and USA.
In phase I, the vaccine is tested among a small number (~20) of healthy adults mainly concerning safety. The vaccine’s properties, and tolerability are tested, and if found appropriate, its clinical laboratory and pharmacological parameters are also recorded.
Gradually, in phase II, the study is expanded. A target population is selected based on characteristics such as age and physical health similar to those for whom the vaccine is intended, to obtain preliminary information on the vaccine’s ability to produce desired effect among them and general safety.
Phase III involves mass testing of thousands. The vaccine is given to a larger number of people to assess protective efficacy and safety yet again. But this time, adequate data needs to be collected so as to clearly show that it is safe and effective for the intended purpose. By the time this stage is reached, vaccine characterization should be complete, that is, virus’ properties should be analysed and how it is related to other viruses should be assessed. Also, final manufacturing process, specifications, and batch release testing procedures must also be ready.
Once clinical trials are done, market authorization can be applied for the vaccine
The data from phase III is used to apply for market authorization at the national regulatory authority in the country. If there are genetically modified organisms, an Environmental Risk Assessment and approval are required. Licensure is when approval is granted to conduct vaccine trial on the general public. Once this process is complete, the vaccine is introduced and used in general population. Both active and passive methods of surveillance are employed to monitor the safety and immunizations post licensure.
COVID-19 Vaccine: 10 candidates in clinical trial stage &123 in preclinical stage
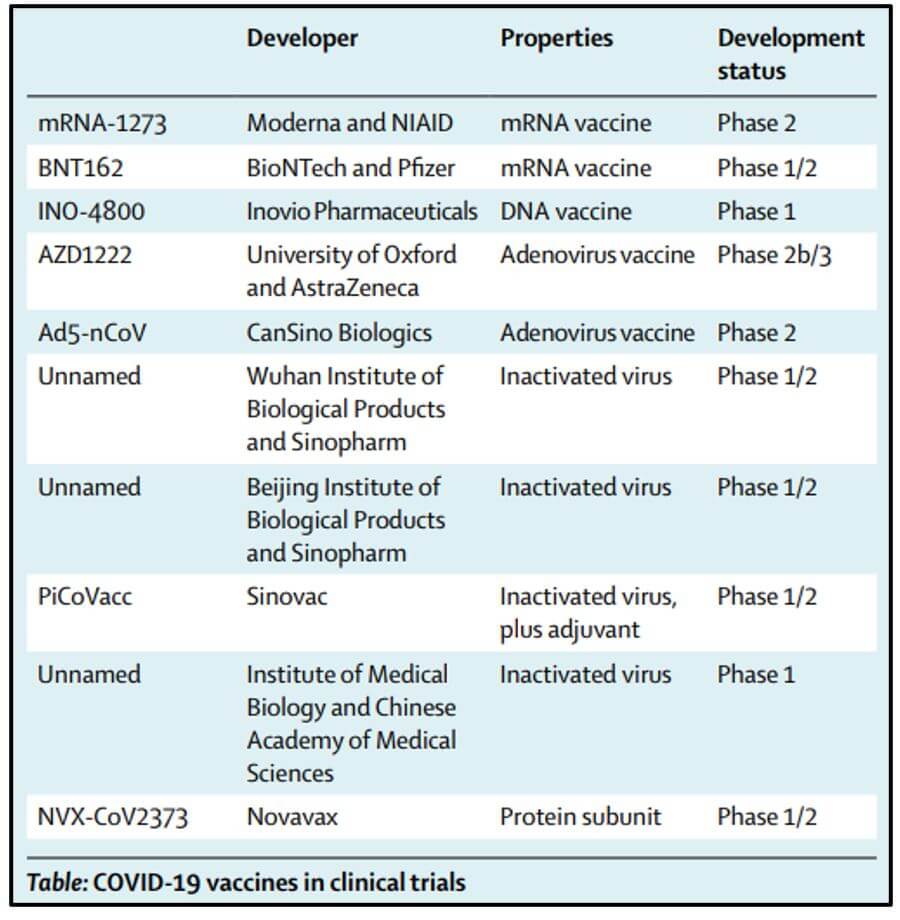
According to WHO’s list of covid-19 vaccine candidates released on 02 June 2020, there are 10 candidate vaccines in the clinic evaluation stage, and 123 in the pre-clinical evaluation stage. Of the ten vaccines in the clinical trial stage, researchers at the University of Oxford and AstraZeneca is in phase III. In phase III, 10,260 subjects will be covered, predominantly from the 18 to 64 years age group. In phase II, the sample size was 1112.
As per a latest report in LANCET, there are seven candidates who are at the phase II of clinical trials. These include CanSino Biological Inc./Beijing Institute of Biotechnology, Moderna/NIAID, Wuhan Institute of Biological Products/Sinopharm, Beijing Institute of Biological Products/Sinopharm, Sinovac, Novavax, and BioNTech/Fosun Pharma/Pfizer. Two candidates are in phase I of vaccine development.
Five candidates out of 123 preclinical trial candidates are from India
Among the 123 candidates at the preclinical stage as of 02 June 2020, the following are from India or have an Indian partner.
- Zydus Cadila
- Bharat Biotech/UW–Madison/FluGen
- Bharat Biotech/Thomas Jefferson University
- Indian Immunologicals Ltd/Griffith University
- Serum Institute of India/Codagenix
The Health Minister had recently said that four out of the 14 COVID-19 vaccine candidates from India may enter the clinical trial stage in the next three-five months.
Prioritization will be key as supply will be limited in the first few months
Even as countries are racing to develop a vaccine for COVID-19, debates have already begun about who will be able to access the vaccine first and who will be given priority. Even if a fast-track approval is given, the supply of the vaccine will be limited in the first few months. The supply may not enough to cater to the demand for a vaccine as large sections of population are affected. There are already calls for prioritization. Some argue that first priority should be given to the frontline healthcare workers working in close proximity with COVID-19 patients. However, if vulnerable individuals are to be given priority, then the older people have to be administered with the vaccine since this age group is highly vulnerable. But this section of the population may need more doses or adjuvants to enhance their immune response.
There are multiple hurdles in the development of a vaccine. Since this is a new virus, the several strains, possible mutations etc. have to be considered while developing the vaccine. Further, there are ethical regulations which cannot not be flouted and safety reasons, that inadvertently cause delay in each stage of the process.
The WHO has warned countries not to depend on herd immunity for managing COVID-19 pandemic. To achieve herd immunity, a certain percentage of population will have to have immunity. Until this immunity level is achieved, there is every possibility that the pandemic will spread, affecting a large population, which may result in the situation going out of control. All this means that the need for vaccine is greater than ever.
Featured Image: COVID-19 Vaccine Development


