A photo is being shared widely on social media with a claim that it shows Dr V. K. Srinivas (Vice President, Bharat Biotech) taking Bharat Biotech’s experimental COVID-19 vaccine (‘COVAXIN’ or BBV152 COVID vaccine) first dose as part of the human clinical trials. The vaccine was developed by Bharat Biotech in collaboration with the Indian Council of Medical Research (ICMR) – National Institute of Virology (NIV). On its official website, Bharat Biotech mentioned that ‘the vaccine received DCGI approval for Phase I & II Human Clinical Trials and the trials will commence across India from July 2020’. Let’s fact-check the claim made in the post.
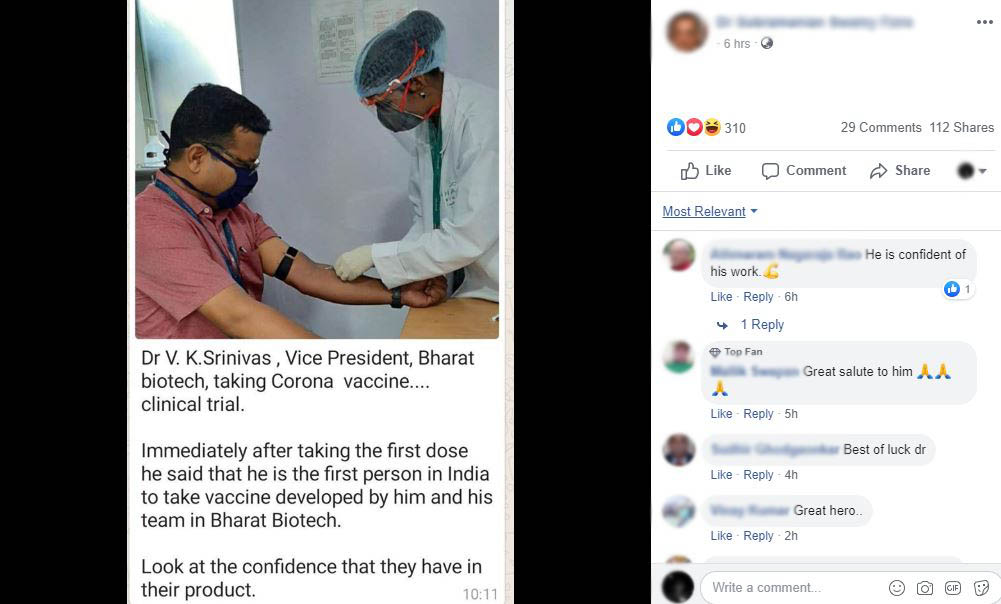
Claim: Photo of Bharat Biotech Vice President taking COVAXIN (indigenous experimental COVID-19 vaccine) first dose.
Fact: Bharat Biotech has clarified that the photo is related to ‘a routine procedural blood draw for testing all production staff’. Also, on the ‘Clinical Trials Registry – India (CTRI)’ website, it can be seen that ‘Date of First Enrollment (India)’ for the BBV152 COVID vaccine begins on 13 July 2020. Hence the claim made in the post is FALSE.
When we searched about the photo, it was found that Bharat Biotech has already clarified regarding the photo. Bharat Biotech has tweeted that the photo is related to ‘a routine procedural blood draw for testing all production staff’.
More details regarding the experimental vaccine can be found on the ‘Clinical Trials Registry – India (CTRI)’ website. On the website, it can be seen that ‘Date of First Enrollment (India)’ for the BBV152 COVID vaccine begins on 13 July 2020. Also, talking to NDTV recently, Dr Krishna Ella (Chairman and MD of Bharat Biotech) said, ‘Most of the clearances are coming through and probably, in 10 days human trials should start’. So, as on 04 July 2020, doses are not given to any human.
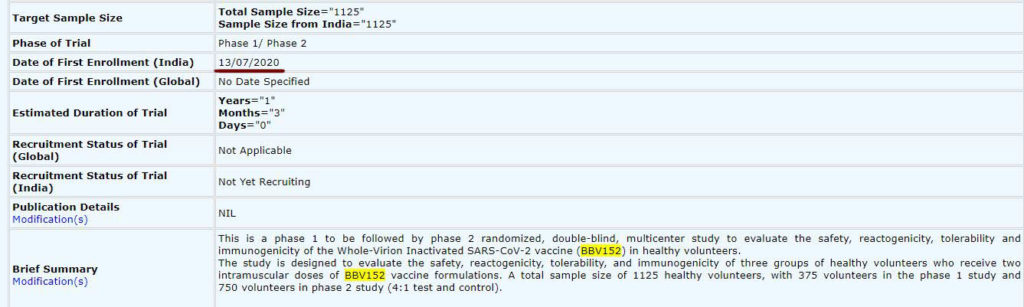
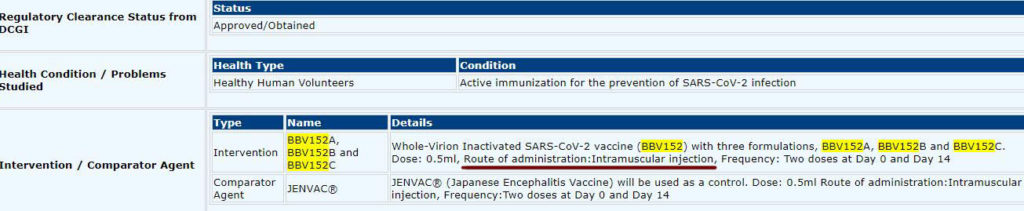
Also, on the CTRI website, it can be read that the healthy volunteers will receive intramuscular doses (‘Route of administration: Intramuscular injection’) of BBV152 vaccine formulations.
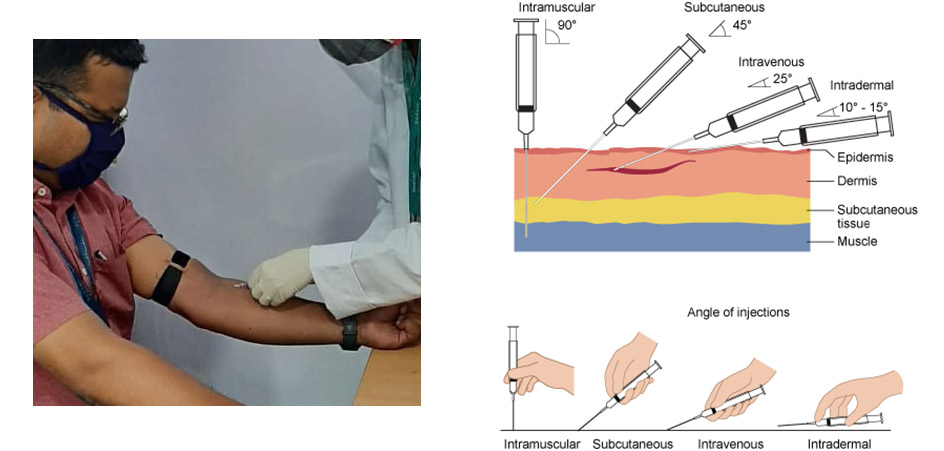
But, the process in the posted photo does not represent intramuscular injection. The difference between various types of injection can be seen in the picture below:
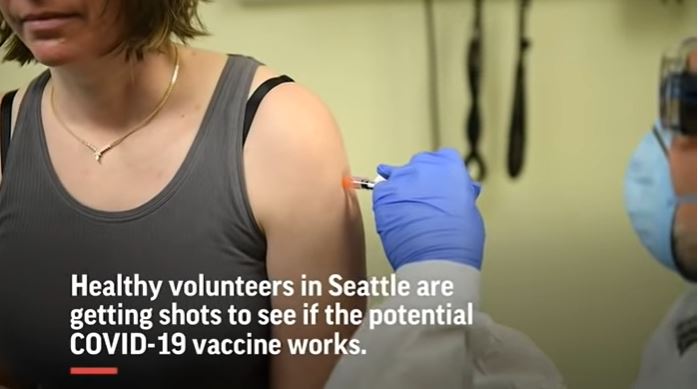
For example, the visuals related to administering of Moderna’s mRNA Vaccine (mRNA-1273) dose (another experimental anti-COVID-19 vaccine) to a woman can be watched here. On the US government’s Clinal Trials website, it can be seen that mRNA-1273 dose is also given as an intramuscular (IM) injection.
To sum it up, the posted photo does not show Bharat Biotech Vice President taking COVAXIN (experimental COVID-19 vaccine) first dose.
Did you watch our new video?

